Producing high-quality medical devices offers a clear benefit to both patients and manufacturers.īut then she saw Travis watching her uneasily, and knew that she had no choice. 
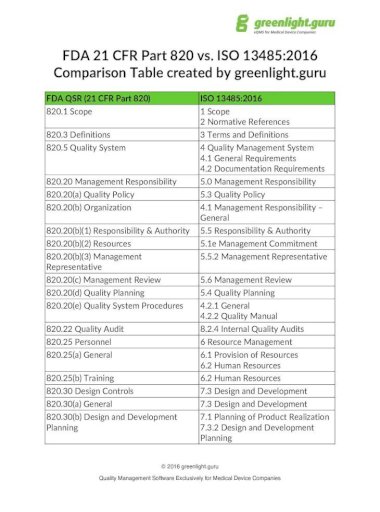
Without a clear set of organizational guidelines, you may accept unnecessary risks of inspection or low-quality products. ISO 13485: How to write a short quality manualĪn electronic quality management system eQMS and a defined process for compliance are critical for medical device companies.ISO is the latest version of this guidance document which outlines the international standards for quality management systems QMS at medical device companies. The ISO has published several international standards documents that relate to medical device manufacturers.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
